Loading...
Loading...
| Jan 01, 1970 | Daily Report |
| RISING BHARAT | News Count (103064) | |
20611. Covaxin demonstrates 93.4% efficacy against severe diseases
Make in India / Atmanirbhar
->
Make in India- Public Health and Infrastructure
English
India
Hindustan Times
|
- Phase 3 clinical trials was an event-driven analysis of 130 symptomatic Covid-19 cases, reported at least two weeks after the 2nd dose.They were conducted at 25 sites across India.
- Bharat Biotech has concluded in the final analysis of the efficacy of the vaccine from phase 3 clinical trials published in a pre-print study.
- According to the study, efficacy data of the whole virion inactivated vaccine further demonstrates 63.6% protection against asymptomatic disease, and 65.2% protection against the Sars-Cov-2, B.1.617.2 Delta variant.
20612. COVID-19 vaccine | Serum Institute plans to double Covishield production in 2022: Report
Make in India / Atmanirbhar
->
Make in India- Public Health and Infrastructure
English
India
Moneycontrol
|
- The Pune-based vaccine manufacturer will scale production of the jab to 200 million doses per month in 2022, from the current 100 million doses a month.
- SII currently has the capacity to manufacture 3 billion doses of vaccines against COVID-19 a year and can raise this to 4 billion doses in 2022, the report said.
- In June, SII also began manufacturing Covovax, Novovax's vaccine against COVID-19.
20613. Government sets up two more labs in Hyderabad, Pune for Covid vaccine testing
Make in India / Atmanirbhar
->
Make in India- Public Health and Infrastructure
English
India
India Tv
|
- These Central Drugs Laboratories have been set up by the Department of Biotechnology at its autonomous research institutes — the National Centre for Cell Science (NCCS), Pune, and the National Institute of Animal Biotechnology (NIAB), Hyderabad
- The country currently has only one Central Drugs Laboratory, at Kasauli in Himachal Pradesh.
- The new labs, set up with funding support from the PM-CARES fund, will carry out batch testing and quality control of vaccines, said a statement issued by the Ministry of Science and Technology.
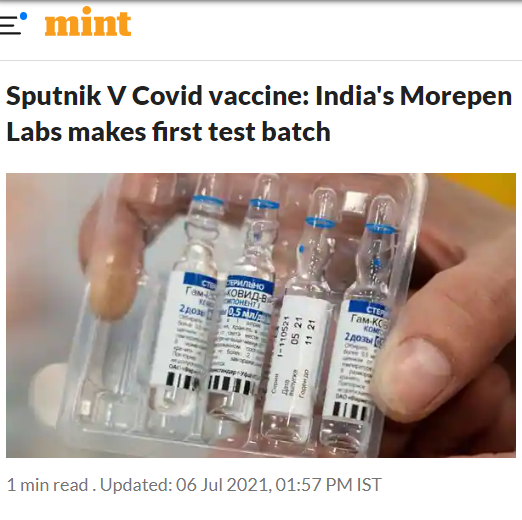
20614. Sputnik V Covid vaccine: India's Morepen Labs makes first test batch
Make in India / Atmanirbhar
->
Make in India- Public Health and Infrastructure
English
India
Mint
|
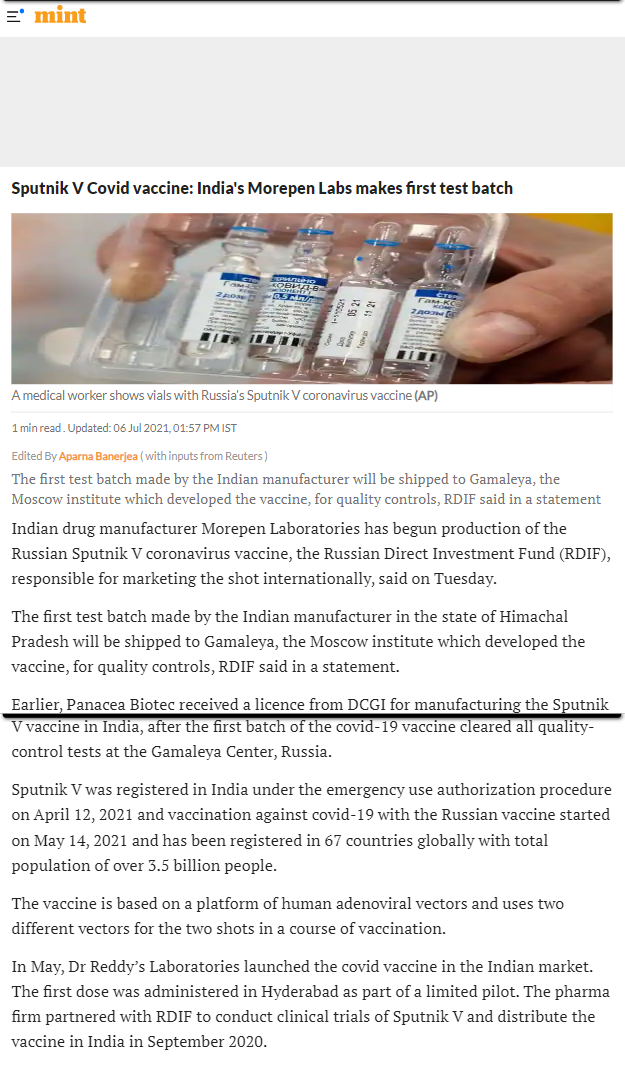
- Indian drug manufacturer Morepen Laboratories has begun production of the Russian Sputnik V coronavirus vaccine, the Russian Direct Investment Fund (RDIF), responsible for marketing the shot internationally
- The first test batch made by the Indian manufacturer will be shipped to Gamaleya, the Moscow institute which developed the vaccine, for quality controls, RDIF said in a statement
- The vaccine is based on a platform of human adenoviral vectors and uses two different vectors for the two shots in a course of vaccination.
20615. Two doses of vaccine give 95% protection against Covid death: ICMR study
India Fights Corona: Vaccination Drive and Initiatives
English
India
Hindustan Times
|
- Two doses of Coronavirus disease (Covid-19) vaccines provide as high as 95% protection against deaths due to Covid-19, and a single dose is 82% effective in preventing deaths, shows the latest study from the Indian Council of Medical Research (ICMR).
- The study that has been published in the Indian Journal of Medical Research (IJMR) was conducted among 117,524 police personnel from the Tamil Nadu police department who either received no shot or at least one Covid-19 vaccine shot
- Tamil Nadu Police department has been documenting vaccination of its members, and Covid-19 deaths during the second wave along with details of the date of hospitalization and vaccination.
20616. India likely to start COVID vaccination for children by September
India Fights Corona: Vaccination Drive and Initiatives
English
India
NEWS SERVICES DIVISION
|
- AIIMS Director Dr Randeep Guleria said, Zydus has already done the trials and they are waiting for the emergency authorization while Bharat Biotech’s Covaxin trials should be over by August or September this year and by that time it should get an approval.
- He said, Pfizer vaccine has already been approved by the US regulator.
- Dr Guleria added that by September, India should start vaccinating children which will be a big boost to break the chain of transmission of COVID-19.
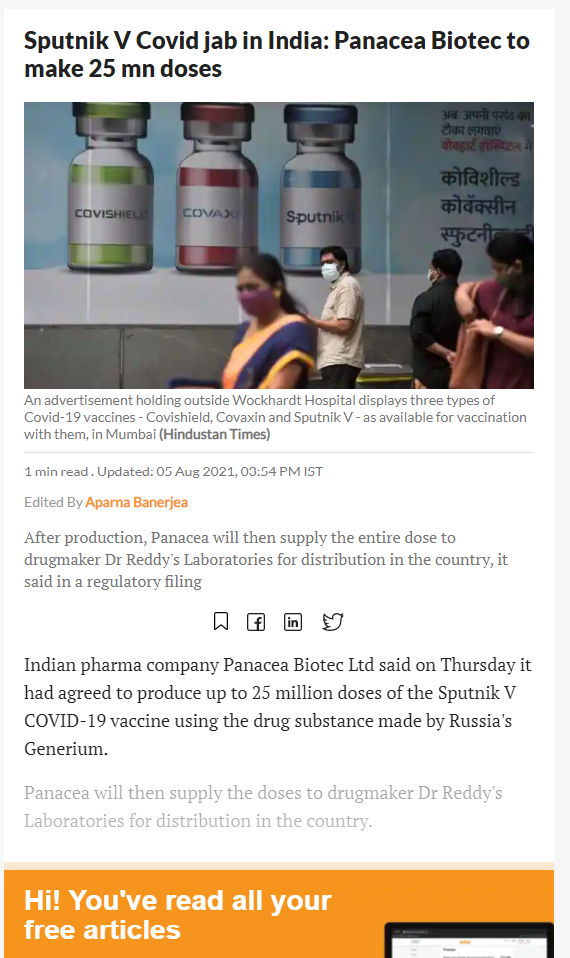
20617. Sputnik V Covid jab in India: Panacea Biotec to make 25 mn doses
Make in India / Atmanirbhar
->
Make in India- Public Health and Infrastructure
English
India
Mint
|
- Indian pharma company Panacea Biotec Ltd said on Thursday it had agreed to produce up to 25 million doses of the Sputnik V COVID-19 vaccine using the drug substance made by Russia's Generium.
- Panacea will then supply the doses to drugmaker Dr Reddy's Laboratories for distribution in the country.
- In September 2020, the company partnered with RDIF to conduct the clinical trials of Sputnik V and distribute the vaccine in India.
- The Hyderabad-based drug major has already stated that the locally manufactured Sputnik V will be available from September-October period.
20618. Covaxin receives GMP certificate from Hungary: Bharat Biotech
Make in India / Atmanirbhar
->
Make in India- Public Health and Infrastructure
English
India
Health World
|
- India's COVAXIN has received Good Manufacturing Practices (GMP) compliance certificate from Hungary.
- So far, more than 18 crore doses administered in the age group of 18-44
- "With this approval Bharat Biotech has achieved another significant milestone in innovating and manufacturing vaccines at Global quality standards and moving forward in the ongoing fight against the COVID-19 pandemic," the note said.
20619. India’s first mRNA Covid-19 vaccine is safe, says expert panel as Centre allows Phase II, III trials
India Fights Corona: Vaccination Drive and Initiatives
English
India
India Today
|
- A vaccine developed usinIg mRNA technology is aimed at training immunity cells to fight covid-19 using genetic instructions.
- The Vccine Experts Committee (VEC) on Tuesday gave its nod to India first mRNA based covid-19 vaccine declaring it safe ,tolerable and immunogenic.
- The US ,FDA (Food and Drug Administration) gave approval to PFozer-BIO nTECH vaccine ,which is also an mRNA-based job. mRNA tecnology is aimed at trainig immunity cells to fight covid-19 using genetic instructions.
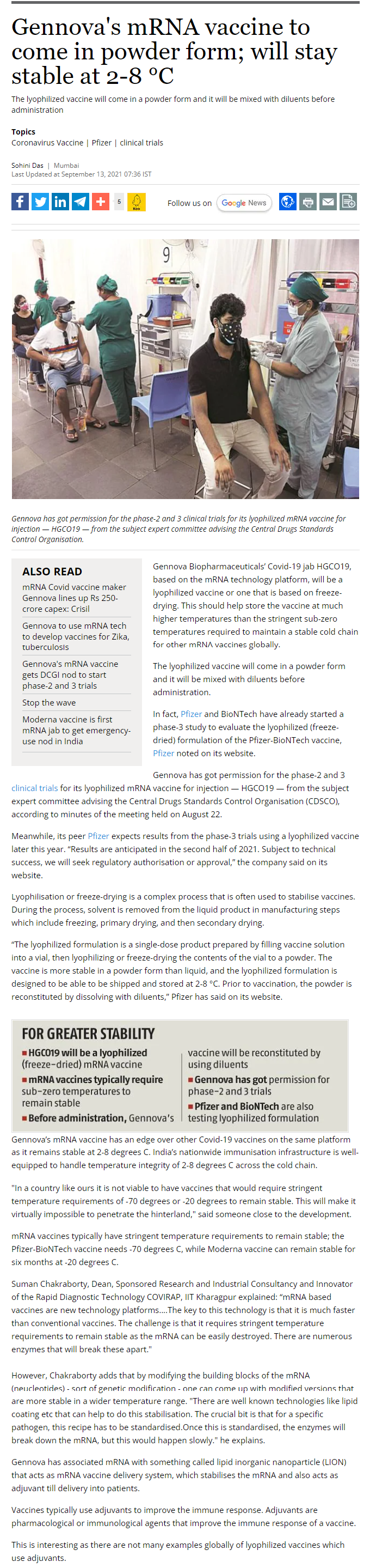
20620. Gennova's mRNA vaccine to come in powder form; will stay stable at 2-8 °C
Make in India / Atmanirbhar
->
Make in India- Public Health and Infrastructure
English
India
Business Standard
|
- Gennova has got permission for the phase-2 and 3 clinical trials for its lyophilized mRNA vaccine for injection — HGCO19 — from the subject expert committee advising the Central Drugs Standards Control Organisation.
- Meanwhile, its peer Pfizer expects results from the phase-3 trials using a lyophilized vaccine later this year. “Results are anticipated in the second half of 2021.
- Gennova’s mRNA vaccine has an edge over other Covid-19 vaccines on the same platform as it remains stable at 2-8 degrees C.
- India’s nationwide immunisation infrastructure is well-equipped to handle temperature integrity of 2-8 degrees C across the cold chain.
- Gennova has associated mRNA with something called lipid inorganic nanoparticle (LION) that acts as mRNA vaccine delivery system, which stabilises the mRNA and also acts as adjuvant till delivery into patients.